Upsalite Climbing Chalk
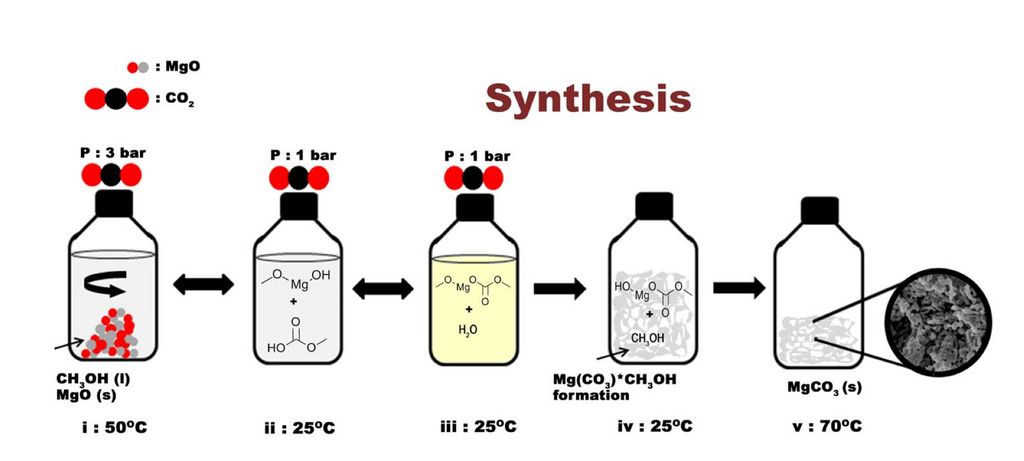
Nanotech is coming to climbing! Climbers use magnesium carbonate to keep their hands dry but magnesium carbonate is used in all kinds of moisture management processes. Magnesium carbonate has an exceptionally high surface area, and an accidental discovery in 2014 at the Nanotech lab in Uppsala, Sweden led to a new form of magnesium carbonate that has a surface area of 800 square meter per gram with insane absorption properties. Mixed in with regular magnesium carbonate this leads to a form of super-chalk for climbers.
It’s a really interesting material worth diving deeper into. Maybe even a good candidate for our own liquid chalk. Who knows?
Abstract, Emphasis Ours: We report the template-free, low-temperature synthesis of a stable, amorphous, and anhydrous magnesium carbonate nanostructure with pore sizes below 6 nm and a specific surface area of ∼ 800 m2 g−1 , substantially surpassing the surface area of all previously described alkali earth metal carbonates. The moisture sorption of the novel nanostructure is featured by a unique set of properties including an adsorption capacity ∼50% larger than that of the hygroscopic zeolite-Y at low relative humidities and with the ability to retain more than 75% of the adsorbed water when the humidity is decreased from 95% to 5% at room temperature. These properties can be regenerated by heat treatment at temperatures below 100°C.The structure is foreseen to become useful in applications such as humidity control, as industrial adsorbents and filters, in drug delivery and catalysis.
